Our research program harnesses the tunable supramolecular chemistry of ionic liquids to engineer biomolecular interfaces that control structure, mechanics, and function across scales. By leveraging how ionic liquids interact with proteins, lipid membranes, and soft biological materials, we develop programmable environments that stabilize biomolecules, modulate cell behavior, and integrate seamlessly with tissues
Research Overview
Research Thrusts
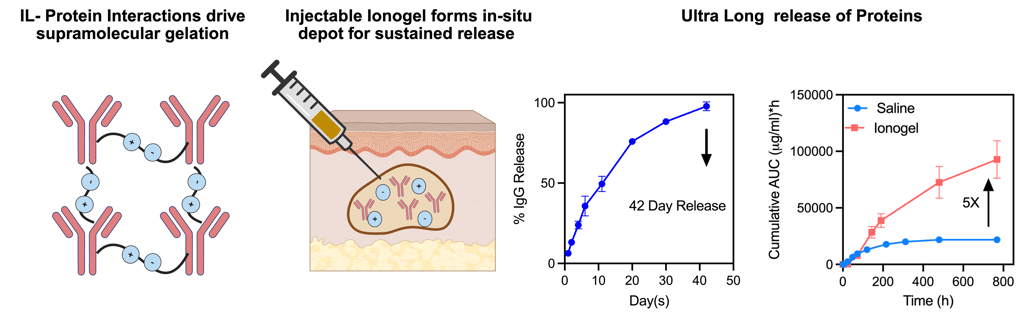
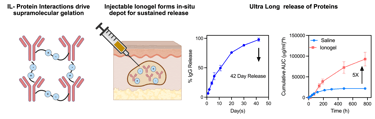
Our group harnesses principles of supramolecular assembly and protein–solute interactions to engineer ionic-liquid–based ionogels capable of sustained, high-fidelity protein delivery. By treating ionic liquids as programmable interfacial environments rather than solvents, we design crosslinker-free depots in which release, stability, and loading capacity emerge from tunable electrostatic pairing, hydrogen bonding, and nanoscale relaxation dynamics. Through multiscale material characterization and disease-specific modeling, we aim to transform how biologics—such as checkpoint inhibitors, enzymes, and cytokines—are administered in settings where long-acting, localized, or ultra-high-concentration delivery is essential.
Ionogels for Long-Acting Protein Delivery
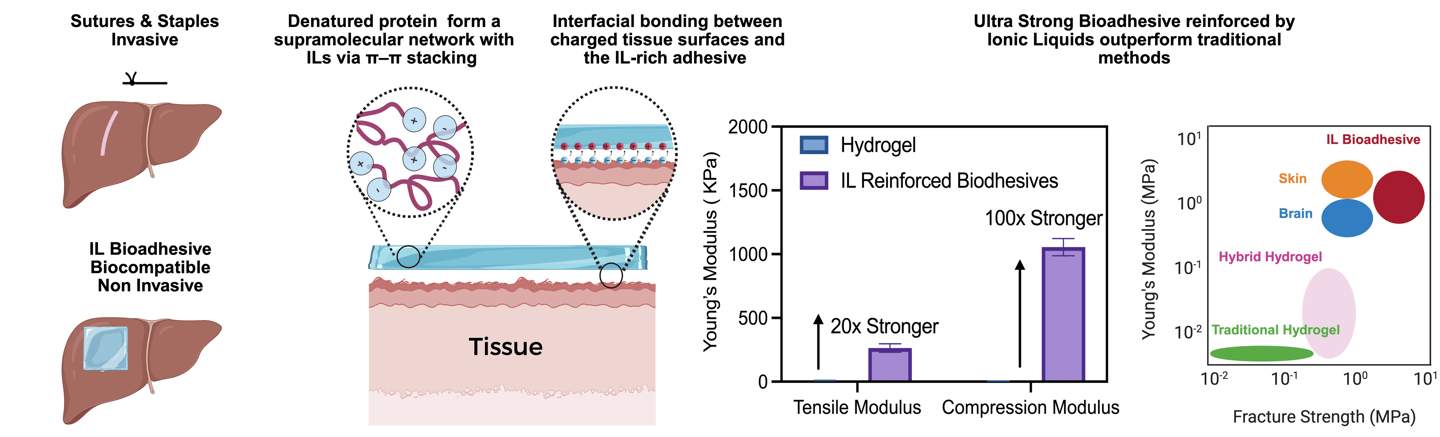
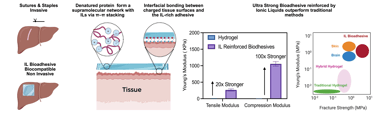
Ionic-Liquid–Reinforced Bioadhesives
We investigate how ionic-liquid–protein interactions can be leveraged to create mechanically robust, wet-compliant, and tissue-integrating bioadhesives. By inducing controlled protein unfolding and supramolecular reinforcement, we generate adhesive networks whose cohesion and interfacial binding are governed by multiscale electrostatic and hydrogen-bonding interactions, rather than covalent chemistry. Our research unites soft-matter mechanics, interfacial science, and protein biophysics to develop a new generation of surgical adhesives that match physiological toughness while supporting regeneration and minimizing trauma.
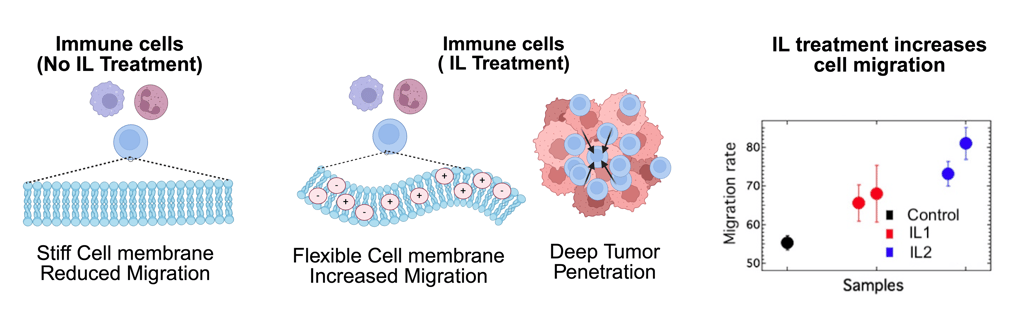
Modulating Membrane Mechanics to Enhance Therapeutic Cell Migration
We apply principles from membrane biophysics and multiscale mechanics to modulate the deformability of immune and stem cells using tailored ionic liquid–lipid interactions. By transiently softening cell membranes—without genetic manipulation or chronic stimulation—we enable cells to navigate dense extracellular matrices, overcome mechanical barriers in solid tumors, and improve homing to injured tissues. Through lipidomics, biophysical profiling, and in vivo functional studies, this thrust establishes a mechanistically grounded framework for engineering cell therapies that operate not only through biochemical signaling, but through optimized physical performance in complex tissue microenvironments.
Contact
Reach out for questions or collaborations
aramesh@pacific. edu
© 2025. All rights reserved.
Office Address
Department of Pharmaceutical Sciences
University of the Pacific
751 Brookside Road
Rm: D11
Stockton
CA 95211
Department of Pharmaceutical Sciences
University of the Pacific
751 Brookside Road
Rm: D4
Stockton
CA 95211
